Deposition (phase transition)
This article needs additional or more specific categories. (January 2025) |
This article includes a list of references, related reading, or external links, but its sources remain unclear because it lacks inline citations. (January 2020) |

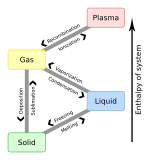
Deposition is the phase transition in which gas transforms into solid without passing through the liquid phase. Deposition is a thermodynamic process. The reverse of deposition is sublimation and hence sometimes deposition is called desublimation.
Applications
[edit]Examples
[edit]One example of deposition is the process by which, in sub-freezing air, water vapour changes directly to ice without first becoming a liquid. This is how frost and hoar frost form on the ground or other surfaces, including leaves. For deposition to occur, thermal energy must be removed from a gas. When the air becomes cold enough, water vapour in the air surrounding a leaf loses enough thermal energy to change into a solid. Even though the air temperature may be below the dew point, the water vapour in the air alone may not condense spontaneously. This supercooled water vapour, when around a leaf, immediately begins to condense. Since it is already past the freezing point, the water vapour changes directly into solid frost.
Soot accumulates as an aerosol deposition. In a fireplace, soot is deposited on the walls of the chimney. Soot particles are aerosolized within volatile organic compounds. These VOCs are in a hot and gaseous state, rising from the fire. When the VOCs come into contact with the walls, they cool, and change to the solid state, without formation of the liquid state. The soot particles also cling to the walls upon contact. This process is made use of industrially in combustion chemical vapour deposition.
Industrial applications
[edit]There is an industrial coating process, known as evaporative deposition, whereby a solid material is heated to the gaseous state in a low-pressure chamber, the gas molecules travel across the chamber space and then deposit to the solid state on a target surface, forming a smooth and thin layer on the target surface. Again, the molecules do not go through an intermediate liquid state when going from the gas to the solid. See also physical vapor deposition, which is a class of processes used to deposit thin films of various materials onto various surfaces.
Deposition releases energy and is an exothermic phase change.
See also
[edit]To From
|
Solid | Liquid | Gas | Plasma |
|---|---|---|---|---|
| Solid | Melting | Sublimation | ||
| Liquid | Freezing | Vaporization | ||
| Gas | Deposition | Condensation | Ionization | |
| Plasma | Recombination |
References
[edit]- Gaja, Shiv P., Fundamentals of Atmospheric Modeling, Cambridge University Press, 2nd ed., 2005, p. 525 ISBN 978-0-521-83970-9
- Moore, John W., et al., Principles of Chemistry: The Molecular Science, Brooks Cole, 2009, p. 387 ISBN 978-0-495-39079-4
- Whitten, Kenneth W., et al., Chemistry, Brooks-Cole, 9th ed., 2009, p. 7 ISBN 978-0-495-39163-0
- Focus on Physical Science. Glencoe Science.